Uganda receives first shipment of long-acting HIV prevention injection lenacapavir
A shipment of 19,200 doses of Lenacapavir, a twice-yearly HIV prevention injection, has arrived in Uganda after regulatory approval. Funded by the Global Fund with U.S. support, distribution begins in March 2026 in high-risk districts, targeting vulnerable groups and strengthening efforts to curb new infections.
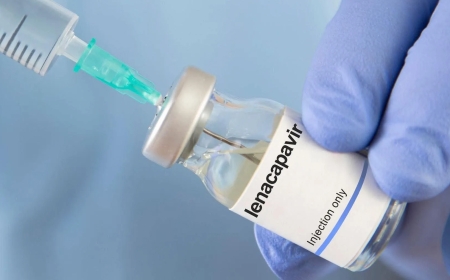
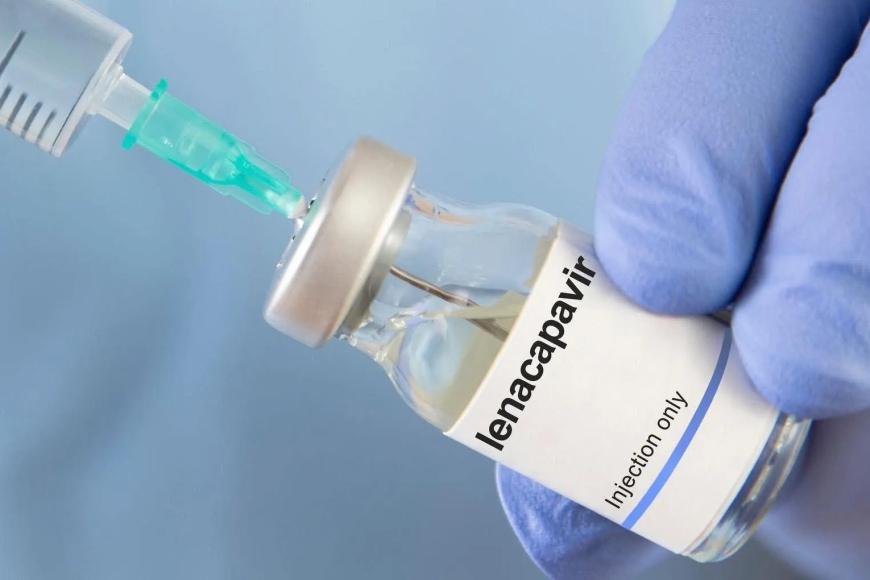
Uganda has received its first shipment of 19,200 doses of Lenacapavir, a long-acting injectable medication used to prevent HIV infection, in what health officials describe as a significant step forward in the country’s ongoing response to the epidemic.
The consignment arrives after approval earlier this year by the National Drug Authority, clearing the way for national rollout. Lenacapavir is administered once every six months and has shown high levels of effectiveness in preventing HIV in clinical trials.
Authorities say the injectable will initially be prioritised for populations at elevated risk of infection, including adolescent girls and young women, who continue to account for a disproportionate share of new HIV cases. The first batch has been financed by the Global Fund to Fight AIDS, Tuberculosis and Malaria, with additional support from the United States government. Distribution is expected to begin in March 2026 in districts recording high HIV incidence.
Health experts believe the twice-yearly injection could strengthen prevention efforts, particularly in communities where adherence to daily oral pre-exposure prophylaxis, or PrEP, has proved challenging. By reducing the need for daily medication, officials hope the long-acting option will improve consistency and uptake among those most at risk.
Uganda’s rollout comes amid broader regional momentum. In neighbouring Kenya, health authorities recently began national distribution of the injectable following regulatory clearance. According to the World Health Organization Regional Office for Africa, Kenya’s launch marked an important milestone, with the first eligible recipients already receiving injections.
Kenya received an initial shipment of 21,000 starter doses through partnerships involving Gilead Sciences, which manufactures Lenacapavir, and the Global Fund. Officials say the drug will be offered free of charge to eligible high-risk individuals at selected public health facilities in priority counties, with further consignments expected in the coming months.
Lenacapavir represents a shift in HIV prevention strategy. Unlike conventional oral PrEP tablets that require strict daily adherence, the biannual injection offers a longer-acting alternative that could ease the burden of daily pill-taking. Clinical trial data cited by global health agencies indicate that the drug significantly reduces the risk of acquiring HIV when used preventively.
In sub-Saharan Africa, where young people remain particularly vulnerable to new infections, health officials see long-acting formulations as a potentially transformative addition to existing prevention tools.
The rollout in Uganda and Kenya has been driven by coordinated global partnerships aimed at expanding access to effective HIV prevention methods in high-burden countries. Public health authorities say the introduction of Lenacapavir could not only lower new infections but also improve engagement with prevention services among communities facing stigma or barriers to care, reinforcing efforts to bring the epidemic under control.


 Kinyarwanda
Kinyarwanda
 English
English
 Swahili
Swahili